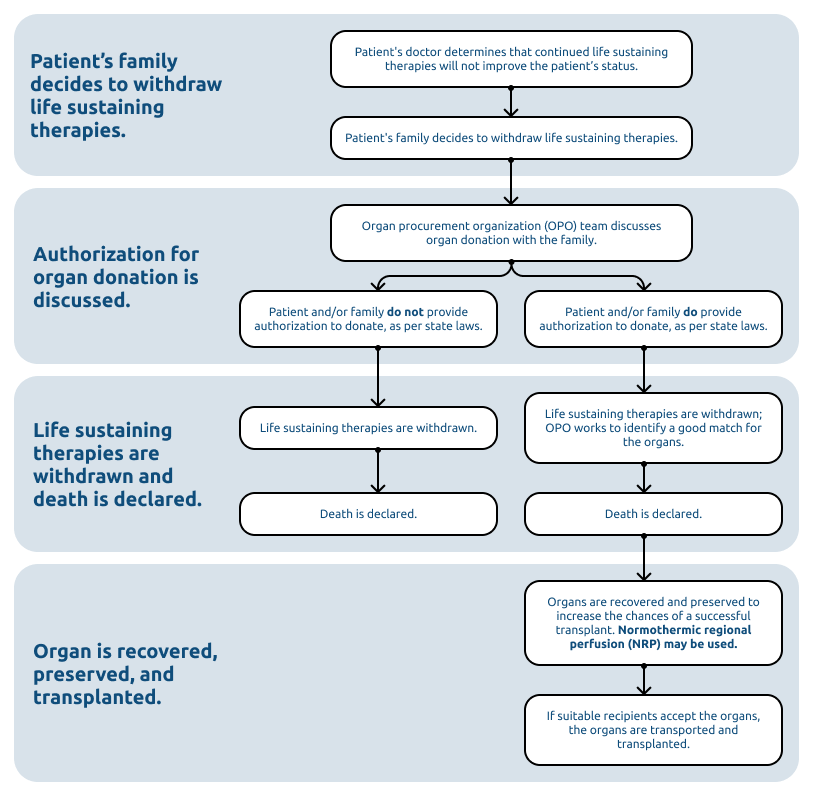
What is NRP? What role does it play in organ donation and transplantation?
Normothermic regional perfusion (NRP) is a technique used after death to help preserve organs intended for transplantation. It can improve the likelihood that organs work properly after transplant and increase the number and quality of organs available for patients in need. Because blood flow and oxygen stop at the time of circulatory death, organs begin to deteriorate quickly, risking their viability for transplant. During NRP, doctors restore warm, oxygen-rich blood to selected organs in the donor’s body using a machine that mimics normal circulation. The circulation established during this process only affects a specific region of the body. By establishing near-normal conditions, NRP helps the organs stay healthier for longer. Organ recovery and NRP are only performed after death has been declared by the patient's medical team, separate from the donation process.
NRP is used during donation after circulatory death (DCD), a type of donation that occurs after a patient’s heart and lungs have permanently stopped functioning and the patient has been pronounced dead by a hospital provider. The flowchart below shows each step in the DCD process and where NRP, if used, fits in.
What can we learn from data?
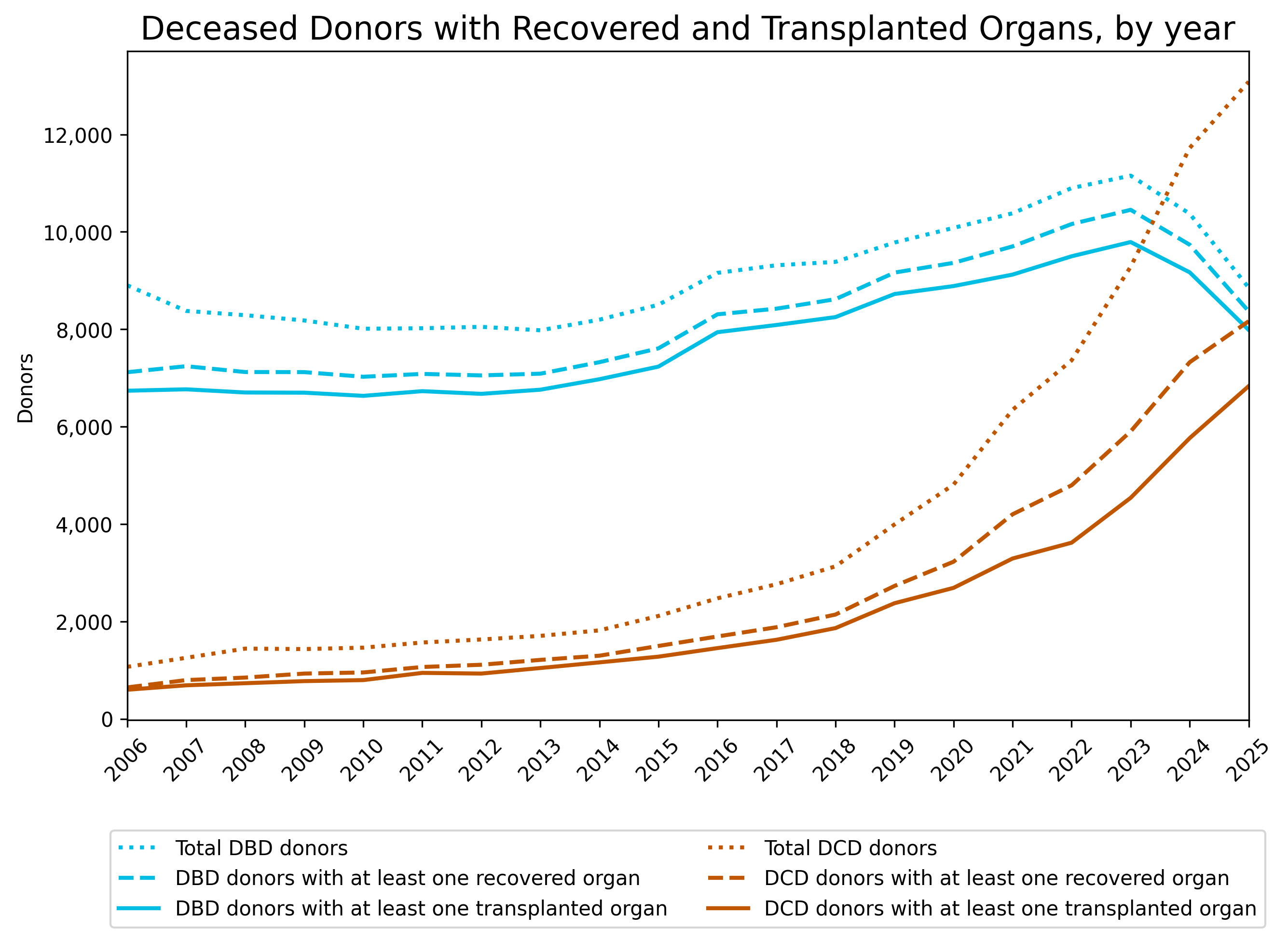
DCD is becoming more common in the United States. In 2025, there were approximately 8,000 DCD donors with at least one recovered organ, comprising 49.2% of all deceased donors with recovered organs, an increase from 42.9% in 2024 and 36.1% in 2023. As DCD grows more frequent, NRP is becoming more regularly used to improve organ viability before transplantation.
How can we ensure safety during NRP?
At the time circulatory death is declared, brain function may vary from patient to patient. There are multiple safeguards to ensure that even if the brain has some function up to the time of death, it will have stopped functioning by the time organs are recovered. These measures include a waiting period after declaration of death to prevent autoresuscitation, and clinical actions such as clamping and venting blood vessels to prevent unintended blood flow to the brain. Policies and procedures around NRP may vary by organ procurement organization (OPO), transplant center, and donor hospital.
Other organizations have published their own statements on or developed recommendations for minimum safety standards in NRP:
Additional statements from organizations and individuals regarding NRP are available on the OPTN’s Ethical Analysis of Normothermic Regional Perfusion Public Comment page.
Donor and recipient safety remain the OPTN’s highest priority. If you would like to report any NRP-related safety concerns, please contact the OPTN. Visit the reporting allegations of misconduct page for more information.
What actions are being taken?
The OPTN, with oversight from HRSA, is working to ensure that NRP is safe and reliable across all OPOs and hospitals at which it is performed.
Progress is already underway:
- Developing safety guidelines for normothermic regional perfusion (NRP): In September 2024, HRSA received a letter concerning potential patient safety risk, ethical issues, and OPTN policy violations during the procurement of organs using NRP. Since then, as directed by HRSA, the OPTN has been developing new guidelines and standards for NRP that ensure donor patient safety. As part of this effort, the President of the OPTN Board issued two letters (letter from 11/6 (PDF - 99 KB), letter from 11/21 (PDF - 98 KB)) to the OPTN community asking programs to commit to specific safety-focused actions. To see all NRP communications between HRSA and the OPTN, please visit the OPTN Critical Comments web page.
- Workgroup focused on safety and family experience: An NRP Workgroup was formed as part of the OPTN Operations & Safety Committee. The workgroup includes representatives from multiple OPTN committees, including, but not limited to, the Patient Affairs Committee (PAC), the Ethics Committee, and the Organ Procurement Organization Committee. The workgroup is focused on safety and family experience for those patients evaluated for potential organ donation including use of NRP and is currently creating new guidelines and standards focused on donor patient safety during NRP. Concurrently, HRSA is continuing to review and provide feedback on the OPTN’s proposed plan to address donor safety during NRP.
- Community Update on NRP: OPTN President John Magee shared an update with the OPTN community on ongoing NRP policy development and reinforces refined safety recommendations to prevent unintended brain reperfusion. His letter highlights procedural safeguards, monitoring, coordination, and transparency, while noting that future policy proposals will incorporate public feedback and undergo formal review. HRSA and the OPTN also developed responses to questions related to NRP from the OPTN community (PDF - 105 KB).
Feedback on this page? Submit it here.